Description
| Product Name | CAT.No | Size |
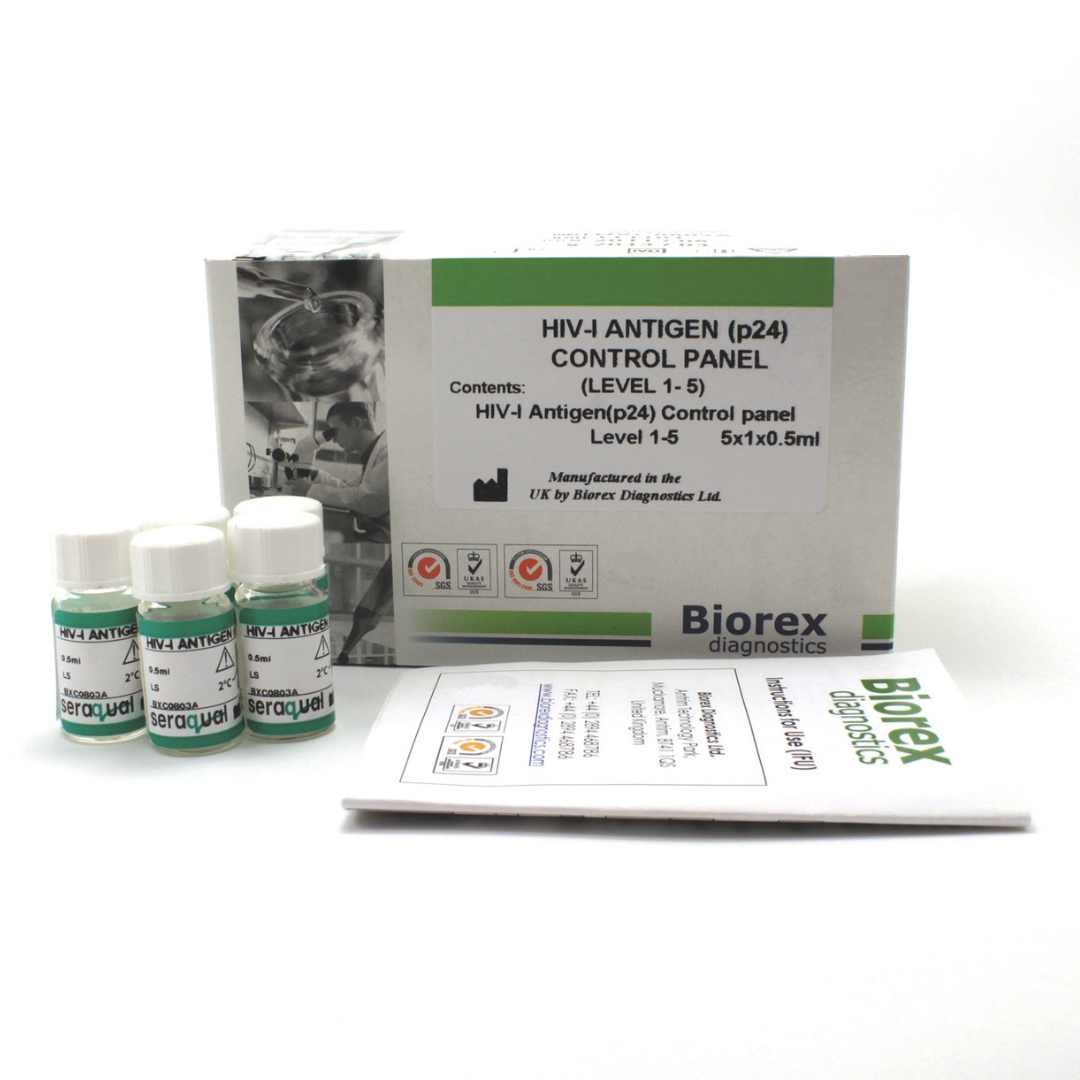
| HIV-I Antigen (p24) Control Panel Level 1 – 4 | BXC0803A | 4 x 1 x 0.5ml |
Product Description
The Biorex Diagnostics HIV-I Antigen Control Panel provides human serum–based, liquid-stable materials for accurate quality control of HIV-I antigen assays. Early detection of HIV-1 infection is critical, particularly during the window period before antibodies are detectable. Reliable laboratory testing depends on precise, reproducible, and clinically relevant controls, and the HIV-I Antigen Control Panel ensures consistent monitoring of assay performance across multiple runs and platforms.
Key Features
- Human serum–based – closely mimics patient samples for clinically relevant testing conditions
- Liquid-stable formulation – ready-to-use for convenient integration into daily laboratory workflows
- Multiple control levels – low, normal, and high antigen concentrations to assess full assay range
- Accurate and reproducible – enables reliable performance monitoring and validation of assay results
- Broad compatibility – suitable for automated, semi-automated, and manual immunoassay systems
- Supports regulatory compliance – aligned with ISO 15189, CAP, and CLIA standards
Benefits
Routine use of the Control Panel allows laboratories to maintain high-quality standards in HIV-1 testing. By providing reliable, human serum–based controls, the panel helps identify deviations in assay performance due to instrument variability, reagent differences, or procedural errors. This ensures accuracy and reproducibility of HIV-I antigen detection, supporting early patient diagnosis, effective monitoring, and informed clinical decision-making.
The control panel is particularly valuable in blood donor screening programmes, reducing the risk of transfusion-transmitted infections and enhancing public health safety. Its multiple control levels allow comprehensive evaluation across the analytical range, ensuring laboratories can confidently assess both low and high antigen concentrations.
Incorporating the Biorex Control Panel into routine laboratory practice strengthens quality assurance, improves laboratory confidence, and ensures compliance with international standards. Designed for both clinical and research applications, this control panel is an essential tool for reliable, reproducible, and accurate HIV-I antigen testing, ultimately supporting better patient outcomes and public health initiatives.
Contact Us for more information.