Description
| Product Name | CAT.No | Size |
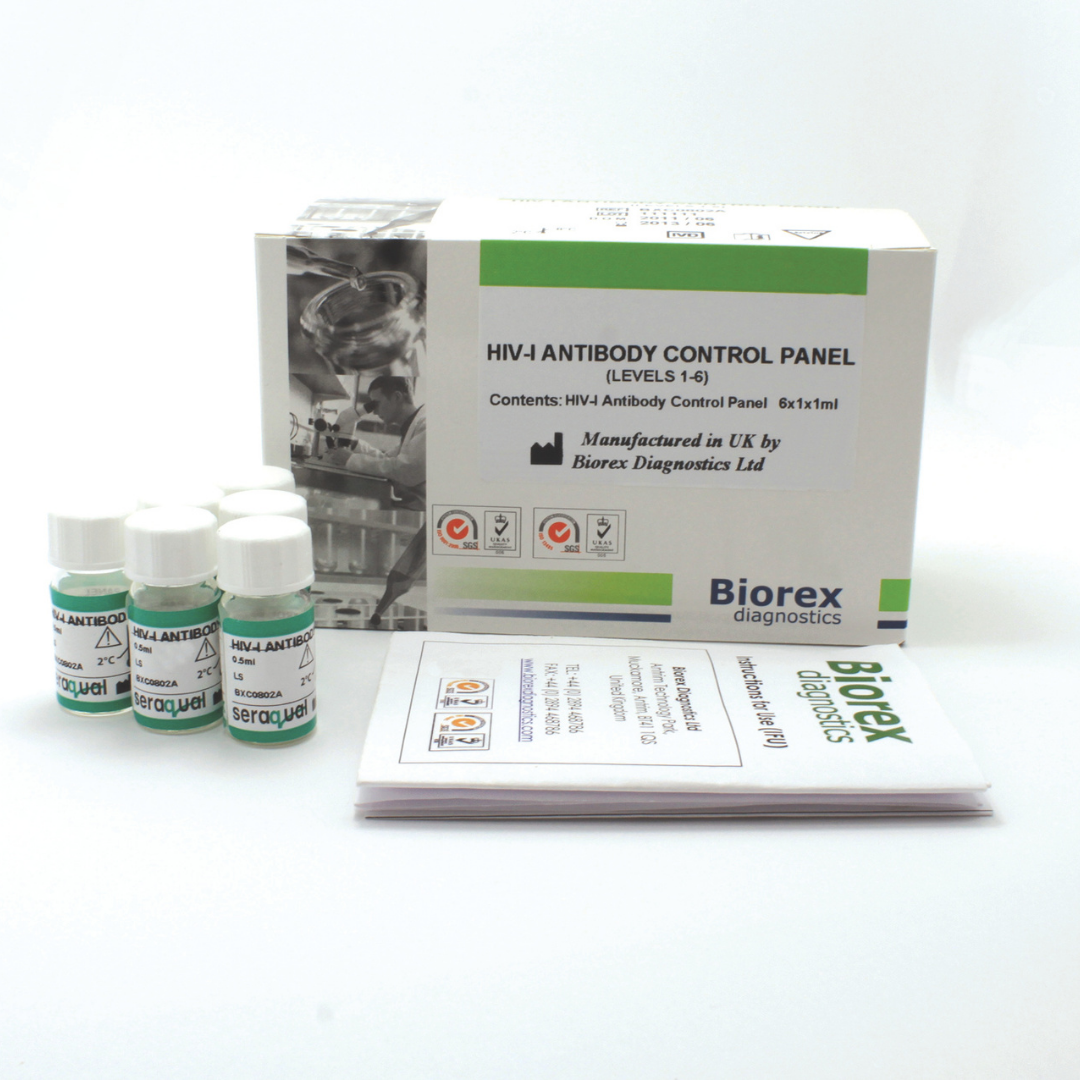
| HIV-I Antibody Control Panel Level 1 – 4 | BXC0802A | 4 x 1 x 1ml |
Product Description
The Biorex Diagnostics HIV-I Control Panel provides human serum–based, ready-to-use materials for accurate quality control of HIV-I antibody assays. Reliable detection of HIV-I antibodies is essential for early diagnosis, patient monitoring, blood donor screening, and public health surveillance. Accurate quality control helps laboratories ensure reproducibility, precision, and reliability in routine testing, supporting timely and informed clinical decision-making.
This control panel is formulated from human serum to closely mimic the composition and behaviour of real patient samples. By reflecting clinically relevant testing conditions, it allows laboratories to assess assay performance across a broad range of HIV-I antibody concentrations. The inclusion of multiple control levels ensures that both low- and high-level antibody responses can be accurately monitored, providing comprehensive quality assurance for diagnostic laboratories.
Key Features
- Human serum-based matrix – simulates patient samples for clinically relevant assay performance.
- Liquid-stable, ready-to-use formulation – simplifies workflow, reduces preparation errors, and ensures consistent results.
- Multiple control levels – low, normal, and high HIV-I antibody concentrations to monitor assay accuracy across the analytical range.
- Reliable and reproducible – enables consistent precision and accuracy assessment in daily testing.
- Compatibility – suitable for use with automated and semi-automated immunoassay systems.
- Regulatory compliance – supports ISO 15189, CAP, and CLIA standards.
Benefits
Routine use of the Biorex HIV-I Control Panel allows laboratories to detect variations in assay performance early, maintain high-quality testing standards, and ensure reproducible results. It supports accurate HIV-I antibody detection for diagnostic purposes, blood screening, and public health monitoring. By providing dependable quality control, laboratories can strengthen their quality assurance programmes, ensure compliance with international standards, and improve clinical confidence in test results. The control panel is an essential tool for laboratories committed to precise, reproducible, and clinically meaningful HIV-I antibody testing, supporting both patient care and research applications.
Contact Us for more information.